Introducing Achieve Life Sciences
Advancing Cytisinicline as the Potential First New Prescription Therapy for Nicotine Dependence in Nearly 20 Years
Achieve Life Sciences, Inc. (Nasdaq: ACHV) is a late-stage specialty pharmaceutical company developing
cytisinicline, a highly selective nicotinic acetylcholine receptor with the potential to become the
standard of care for both smoking and vaping cessation. With a strong clinical foundation, a
near-term regulatory milestone, and an accelerated path for a second indication, Achieve is
positioned to transform treatment for a large, underserved market.
Approximately 29 million adults in the United States smoke combustible cigarettes, and about 7
million attempt to quit each year. In addition, around 17 million U.S. adults use e-cigarettes or
vaping, yet there are no FDA-approved treatments specifically indicated to support nicotine e-
cigarette cessation.
Cytisinicline is a plant-based alkaloid with a high binding affinity to the nicotinic acetylcholine
receptor. It is believed to aid in treating nicotine addiction for smoking and e-cigarette cessation
by interacting with nicotine receptors in the brain, reducing the severity of nicotine craving
symptoms, and reducing the reward and satisfaction associated with nicotine products.
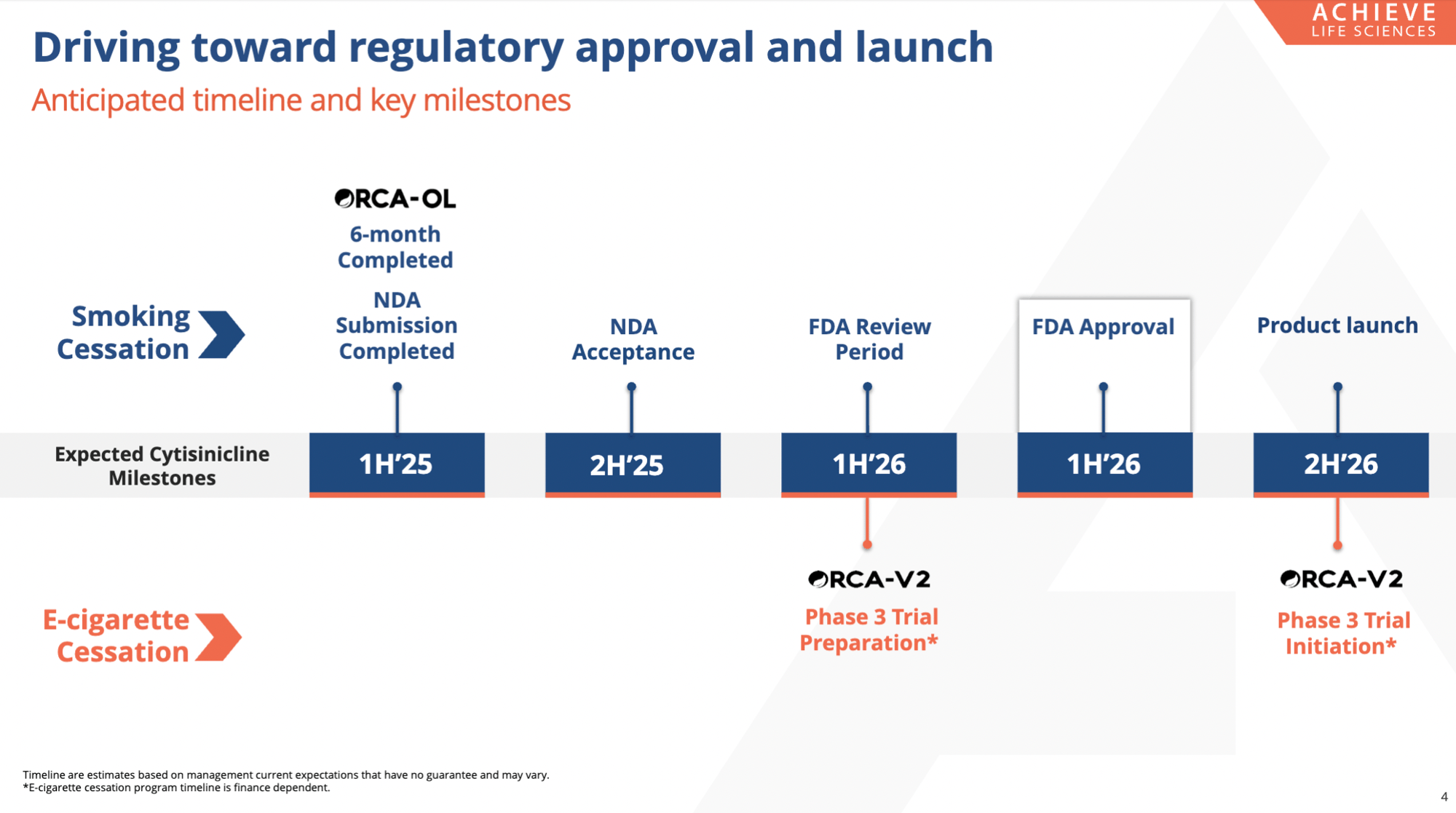
Achieve’s cytisinicline NDA for the treatment of nicotine dependence for smoking cessation is
under FDA review, with a PDUFA date of June 20, 2026. Additionally, the vaping indication
received the FDA’s first CNPV, which enables an accelerated development pathway and an
expedited approval process. Achieve anticipates initiating a single Phase 3 trial in the first half of
2026.
~480K
Deaths in the U.S. Annually
1.1B
Global Smokers
$300B
Annual Healthcare Costs Associated with Smoking Costs in the U.S.
Recent Announcements
ORCA Development Program